Atomic StructureHard
Question
A mixture of 2 moles of helium gas (atomic mass = 4 amu) and 1 mole of argon gas (atomic mass = 40 amu) is kept at 300 K in a container. The ratio of the rms speeds 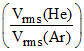 is
is
Options
A.0.32
B.0.45
C.2.24
D.3.16
More Atomic Structure Questions
The Kα X-ray emission line of tungsten occurs at λ = 0.021 nm. The energy difference between K and L levels in...An alphanucleus of energy 1/2 mv2 bombards a heavy nuclear target of charge Ze. Then the distance of closest approach fo...A gas is compressed isothermally to half its initial volume. The same gas is compressed separately throughan adiabatic p...Beta rays emitted by a radioactive material are...If the binding energy per nucleon of a nuclide is high then...