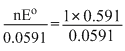ElectrochemistryHard
Question
The standard e.m.f of a cell, involving one electron change is found to be 0.591 V at 25oC. The equilibrium constant of the reaction is (F = 96,500 C mol-1: R = 8.314 JK-1 mol-1)
Options
A.1.0 × 101
B.1.0 × 1030
C.1.0 × 1010
D.1.0 × 105
More Electrochemistry Questions
Through a solution of CuSO4 a current of 3 amperes was passed for 2 hours. At cathode 3 g of Cu2+ ions were discharged. ...Given EoCr3+/Cr = - 0.72V, EoFe2+/Fe = - 0.42 V. The potential for the cell Cr | Cr3+ (0.1M)||Fe2+(0.01M)|Fe is...An increase in equivalent conductance of a strong electrolyte with dilution is mainly due to...Sulfide ores are common for the metals -...Amount of oxalic acid present in a solution can be determined by its titration with KMnO4 solution in the presence of H2...