ElectrochemistryHard
Question
An increase in equivalent conductance of a strong electrolyte with dilution is mainly due to
Options
A.Increasing in ionic mobility of ions
B.100% ionisation of electrolyte at normal dilution
C.increase in both , ie, number of ions and ionic mobility of ions
D.increase in number of ions
Solution
λeq = k × V = 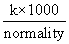
On dilution, the number of current carrying particles per cm decreases but the volume of solution increases. Consequently, the ionic solution increases, which on turns increases the equivalent conductance of strong electrolyte.
On dilution, the number of current carrying particles per cm decreases but the volume of solution increases. Consequently, the ionic solution increases, which on turns increases the equivalent conductance of strong electrolyte.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Given :E0(Cr+3/Cr) = -0.74 ; E0(MnO4-/Mn+2) = 1.51 VE0(Cr2O7-2/Cr+3) = 1.33 V; E0(Cl/Cl-) = 1.36 VBased on the data give...For a reaction A(s) + 2B+ → A2+ + B(s); KC has been found to be 1012. The EMF of the cell is...Which of the following curve represents the variation of λM with √C for AgNO3 ?...A cell whose resistance, when filled with 0.1 M – KCl is 200 Ω, is measured to be 6400 Ω, when filled with 0.003 M – NaC...The EMF of the cell: Hg(l)|Hg2Cl2(s), KCl sol. (1.0N)|Quinohydrone|Pt, is 0.210 V at 298 K. What is the pH of the quinoh...