ElectrochemistryHard
Question
Through a solution of CuSO4 a current of 3 amperes was passed for 2 hours. At cathode 3 g of Cu2+ ions were discharged. The current efficiency is [At. wt. of Cu = 63.5]
Options
A.33.3%
B.42.2%
C.48.7%
D.54.4%
Solution
According to law of electrolysis, Mass deposited (m) - Z i t
or i =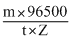
Here, m = 3g, t = 2 t = 2 × 60 × 60 = 7200sec
Z =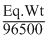 ; Eq. wt =
; Eq. wt = 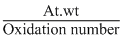
∴ i =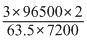 = 1.266A
= 1.266A
Efficiency of current
= × 100
× 100
=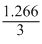 × 100 = 42.22%
× 100 = 42.22%
or i =
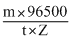
Here, m = 3g, t = 2 t = 2 × 60 × 60 = 7200sec
Z =
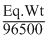 ; Eq. wt =
; Eq. wt = 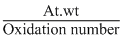
∴ i =
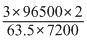 = 1.266A
= 1.266A Efficiency of current
=
 × 100
× 100=
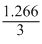 × 100 = 42.22%
× 100 = 42.22%Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
The value of equilibrium constant for a feasible cell reaction must be...For the cell Zn|Zn2+||Cu2+|Cu, if the concentration of both, Zn2+ and Cu2+ ions are doubled, the EMF of the cell...F2 gas cannot be obtained by the electrolysis of any aqueous fluoride salt because...For Zn2+ / Zn, Eo = - 0.76 V, for Ag+/Ag Eo = 0.799 V. The correct statement is -...Which of the following molecule contains contains one lone pair of electrons on the central atom?...