SolutionHard
Question
Benzene and toluene form nearly ideal solutions. At 20oC, the vapour pressure of benzene is 75 torr and that of toluene is 22 torr. The partial vapour pressure of benzene at 20oC for a solution containing 78 g of benzene and 46 g of toluene in torr is
Options
A.50
B.25
C.37.5
D.53.5
More Solution Questions
pH of a 0.1 M monobasic acid is found to be 2. Hence its osmotic pressure at a given temp. T K is-...Mole fraction of a non-electrolyte in aqueous solution is 0.07. If Kf is 1.86o mol-1 kg, depression in f.p., ᐃTf, ...For a dilute solution, Raoult′s law states that :...An electrolysis cell contains a solution of Ag2SO4 and has platinum electrodes. A current is passed until 1.6 gm of O2 h...Assuming the formation of an ideal solution, determine the boiling point of a mixture containing 1560 g benzene (molar m...
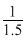 = 50 torr
= 50 torr