SolutionHard
Question
Mole fraction of a non-electrolyte in aqueous solution is 0.07. If Kf is 1.86o mol-1 kg, depression in f.p., ᐃTf, is:
Options
A.0.26o
B.1.86o
C.0.13o
D.7.78o
Solution
Firstly we have to convert mole fraction into molality.
Molality =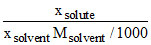
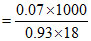 = 4.18
= 4.18
Now, ᐃTf = kf m
= 1.86 × 4.18 = 7.780 .
Molality =
Now, ᐃTf = kf m
= 1.86 × 4.18 = 7.780 .
Create a free account to view solution
View Solution FreeMore Solution Questions
At a constant temperature, DS will be maximum for which of the following processes :...A very small amount of non-volatile solute (that does not dissociate) is dissolved in 56.8 cm3 of benzene (density 0.889...If vapour pressures of pure liquids ′A′ & ′B′ are 300 and 800 torr respectively at 25oC. When th...When 36.0 g of a solute having the empirical formula CH2O is dissolved in 1.20 kg of water, the solution freezes at −0.9...At 100oC the vapour pressure of a solution of 6.5 g of a solute in 100 g water is 732 mm. If Kb = 0.52, theboiling point...