SolutionHard
Question
Assuming the formation of an ideal solution, determine the boiling point of a mixture containing 1560 g benzene (molar mass = 78) and 1125 g chlorobenzene (molar mass = 112.5) using the following against an external pressure of 1000 Torr.
Options
A.90oC
B.100oC
C.110o
D.120oC
Solution
20 mole C6H6, 10 mole C6H5Cl ⇒ XB = 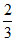 , XC =
, XC = 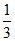
at t = 100oC ⇒ ps = 300 ×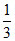 + 1350 ×
+ 1350 × 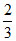 = 100 + 900 (=1000).
= 100 + 900 (=1000).
at t = 100oC ⇒ ps = 300 ×
Create a free account to view solution
View Solution FreeMore Solution Questions
How many moles of sucrose should be dissolved in 500 gms of water so as to get a solution which has a difference of 104o...Van′t Hoff factor is :...A solution of x moles of sucrose in 100 grams of water freezes at -0.2oC. As ice separates the freezing point goes down ...An aqueous solution of 0.01 M CH3COOH has Van′t Hoff factor 1.01. If pH = - log [H+], pH of 0.01 M CH3COOH solutio...FeCl3 solution (side X) on reaction with K4[Fe(CN)6] (side Y) in aqueous solution gives blue colour. These are separated...