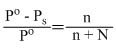SolutionHard
Question
For a dilute solution, Raoult′s law states that :
Options
A.the lowering of vapour pressure is equal to the mole fraction of solute
B.the relative lowering of vapour pressure is equal to the mole fraction of solute
C.the relative lowering of vapour pressure is proportional to the amount of solute in solution
D.the vapour pressure of the solution is equal to the mole fraction of solvent
More Solution Questions
The process of getting fresh water from sea water is known as...The amount of ice that will separate out on cooling a solution containing 50 g of ethylene glycol in 200 g water to - 9....What is the normal boiling point of the solution represented by the phase diagram ?...When 20 g of naphthoic acid (C11H8O2) is dissolved in 50 g of benzene (Kf = 1.72 K kg mol-1), a freezing point depressio...Moles of K2SO4 to be dissolved in 12 mol water to lower its vapour pressure by 10 mmHg at a temperature at which vapour ...