ElectrochemistryHard
Question
Resistance of a conductivity cell filled with a solution of an electrolyte of concentration 0.1 M is 100 Ω. The conductivity of this solution is 1.29 S m-1. Resistance of the same cell when filled with 0.2 M of the same solution is 520 Ω. The molar conductivity of 0.02 M solution of the electrolyte will be
Options
A.124 × 10-4S m2 mol-1
B.1240 × 10-4 S m2 mol-1
C.1.24 × 10-4 S m2 mol-1
D.12.4 × 10-4 S m2 mol-1
Solution
There is one mistake in Question paper.
Assuming concentration of solution is 0.2 M instead of 0.02 M. Since resistance of 0.2 M is 520 Ω.
R = 100 Ω
K =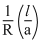
1.29 =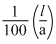
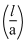 = 129 m-1
= 129 m-1
R = 520Ω , C = 0.2 M
K =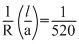 (129) Ω-1 m-1
(129) Ω-1 m-1
μ = K × Vin cm3
=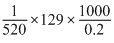 × 10-6
× 10-6
= 1.24 × 10-3
= 12.4 × 10-4
Assuming concentration of solution is 0.2 M instead of 0.02 M. Since resistance of 0.2 M is 520 Ω.
R = 100 Ω
K =
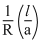
1.29 =
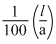
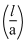 = 129 m-1
= 129 m-1R = 520Ω , C = 0.2 M
K =
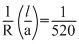 (129) Ω-1 m-1
(129) Ω-1 m-1μ = K × Vin cm3
=
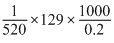 × 10-6
× 10-6= 1.24 × 10-3
= 12.4 × 10-4
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Standard reduction potentials of the half reactions are given below: F2(g) + 2e- → 2F-(aq) ; Eo = + 2.85 V Cl2(g) ...The oxidation number of Cr in K2Cr2O7 is :...ᐃG = ᐃH - TᐃS and ᐃG = ᐃH + T then is :...Three moles of electrons are passed through three solutions in succession containing AgNO3, CuSO4, and AuCl3, respective...During an electrolysis of conc. H2SO4, perdisulphuric acid (H2S2O8) and O2 form in equimolar amount. The amount of H2 th...