ElectrochemistryHard
Question
During an electrolysis of conc. H2SO4, perdisulphuric acid (H2S2O8) and O2 form in equimolar amount. The amount of H2 that will form simultaneously at other electrode will be (2H2SO4 → H2S2O8 + 2H+ + 2e-)
Options
A.thrice that of O2 in moles
B.twice that of O2 in moles
C.equal to that of O2 in moles
D.half of that of O2 in moles
Solution
Anode 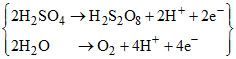
Cathode {2H2O → H2 + 2OH- - 2e-} × 3.
__________________________________
Net : 2H2SO4 + 8H2O → H2S2O8 + O2 + 3H2 + 6H+ + 6OH-
Hence ratio of and is 1 : 3.
Cathode {2H2O → H2 + 2OH- - 2e-} × 3.
__________________________________
Net : 2H2SO4 + 8H2O → H2S2O8 + O2 + 3H2 + 6H+ + 6OH-
Hence ratio of and is 1 : 3.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Pure water is saturated with pure solid AgCl, a silver electrode is placed in the solution and the potential is measured...In hydrogen - oxygen fuel cell, combustion of hydrogen occurs to...Electrolysis of a solution of MnSO4 in aqueous sulphuric acid is a method for the preparation of MnO2 as per the reactio...The efficiency of a fuel cell is 80% and the standard heat of reaction is -300kJ. The reaction involves two electrons in...The standard reduction potentials in acidic conditions are 0.77 V and 0.53 V, respectively, for Fe3+|Fe2+ and I3−|I− cou...