ElectrochemistryHard
Question
ᐃG = ᐃH - TᐃS and ᐃG = ᐃH + T 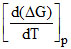 then
then 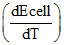 is :
is :
Options
A.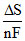
B.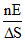
C.- nFEcell
D.+ nEFcell
More Electrochemistry Questions
The standard reduction potential for the reactions: Ag+ + e− → Ag and Ag(NH3)2+ + e− → Ag + 2NH3 are +0.79 V and +0.37 V...Arrange following compounds in decreasing order of electrophilic substitution. (i) (ii) (iii) (iv)...Which of the following statements is true for the electrochemical, Daniel cell ?...The voltage of the cell given below is −0.61 V. Pt|H2 (1 bar)|NaHSO3 (0.4 M), Na2SO3 (6.4 × 10−2 M)||Zn2+ (0.4 M)|Zn If ...The quantity of electricity required for the reduction of 1 mole of Fe2O3 to Fe is...