ElectrochemistryHard
Question
Three moles of electrons are passed through three solutions in succession containing AgNO3, CuSO4, and AuCl3, respectively. The molar ratio of amounts of cations reduced at cathode will be
Options
A.1 : 2 : 3
B.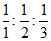
C.3 : 2 : 1
D.6 : 3 : 2
Solution
Ag Cu Au
equivalent 1 : 1 : 1
ratio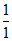 :
: 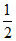 :
: 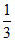
6 : 3 : 2.
equivalent 1 : 1 : 1
ratio
6 : 3 : 2.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Four colourless salt solutions are placed in separate test tubes and a strip of copper is placed in each. Which solution...Rust is a mixture of...Consider the reaction : (T = 298 K)Cl2(g) + 2Br-(aq) → 2Cl- (aq) + Br2 (aq.)The emf of the cell, when [Cl-] = [Br2...The EMFs of the cell obtained by combining separately Zn and Cu electrodes of a Daniel cell with normal calomel electrod...Consider the following Galvanic cell as shown in figure. By what value the cell voltage change when concentration of ion...