ElectrochemistryHard
Question
The oxidation number of Cr in K2Cr2O7 is :
Options
A.+ 3
B.- 3
C.+6
D.-6
Solution
oxidation number of Cr in K2 Cr2O7
2 + 2 × x - 2 × 2 = 0
x =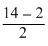 = + 6
= + 6
2 + 2 × x - 2 × 2 = 0
x =
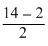 = + 6
= + 6Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
F2 gas cannot be obtained by the electrolysis of any aqueous fluoride salt because...What is the pH of 0.01 m glycine solution ? for glycine, Ka1 = 4.5 × 10-3 and Ka2 = 1.7 × 10-10 at 298K ?...An aqueous solution of X is added slowly to an aqueous solution of Y as shown in List-I. The variation in conductivity o...For a electrochemical cell Zn|Zn2+ (C1 M)||Cu2+ (C2 M)|Cu, the decrease in free energy at a given temperature is a funct...Select the correct statement.(i) Delocalisation of σ-electron is hyperconjugation.(ii) Delocalisation of π-el...