ThermodynamicsHard
Question
5.6 liter of helium gas at STP is adiabatically compressed to 0.7 lit. Taking the initial temperaure to be T1, the work done in the process is
Options
A.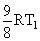
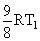
B.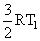
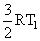
C.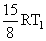
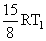
D.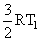
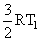
Solution
TVγ-1 = C
T2 (0.7)2/3 ⇒ T2 = T1(8)2/3 = 4T1
∴ᐃw (work done on the system)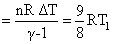
T2 (0.7)2/3 ⇒ T2 = T1(8)2/3 = 4T1
∴ᐃw (work done on the system)
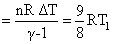
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Steam at 100o C is passes into 1.1 kg of water contained in a calorimeter of water equivalent 0.02 kg at 15oC till the t...During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio...Two moles of helium gas are taken over the cycle ABCDA, as shown in the P - T diagram. Assuming the gas to be ideal the ...In an adiabatic process the quantity which remains constant is :...Two moles of ideal helium gas are in a rubber balloon at 30oC. The balloon is fully expandable and can be assumed to req...