ThermodynamicsHard
Question
Two moles of helium gas are taken over the cycle ABCDA, as shown in the P - T diagram.
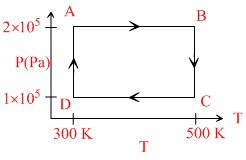
Assuming the gas to be ideal the work done on the gas in taking it from A to B is

Assuming the gas to be ideal the work done on the gas in taking it from A to B is
Options
A.200 R
B.300 R
C.400 R
D.500 R
Solution
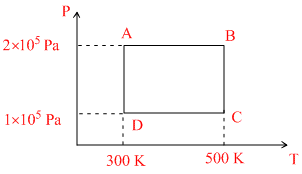
WAB = ᐃQ - ᐃU = nCpdT - nCvdT (at constant pressure)
= n(Cp - Cv)dt
= nRdT = 2 × R × (500 - 300) = 400 R
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A quantity of heat requirad to change the unit mass of a solid substance to its liquid state, while the temperature rema...In the following $p - V$ diagram the equation of state along the curved path is given by $(V - 2)^{2} = 4ap$ where $a$ i...A gas speciman in one vessel is expended isothermally to double its volume and a simillar specimen in the second vessel ...5.6 liter of helium gas at STP is adiabatically compressed to 0.7 lit. Taking the initial temperaure to be T1, the work ...1 calorie is equal to how many joules ?...