NEET | 2013ThermodynamicsHard
Question
During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of Cp/Cv for the gas is :-
Options
A.3/2
B.4/3
C.2
D.5/3
Solution
P ∝ T3 and PV = nRT gives PV3/2 = constant
⇒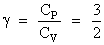
⇒
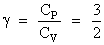
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
A gas at intial temperature 27oC is adiabatic cally compressed so that its volume becomes 1 / 9 of the previous one. If ...The ratio of the speed of sound in nitrogen gas to that in helium gas, at 300 K is...If 10 gm ice at 0oC is mixed with 10 gm water at 20oC, the final temperature will be :-...An ideal gas expands in such a way that PV2 = constant throughout the process. Select correct alternative...In an isometric change :...