ThermodynamicsHard
Question
Two moles of ideal helium gas are in a rubber balloon at 30oC. The balloon is fully expandable and can be assumed to require no energy in its expansion. The temperature of the gas in the balloon is slowly changed to 35oC. The amount of heat required in raising the temperature isnearly (take R = 8.31 J/mol.K)
Options
A.62 J
B.104 J
C.124 J
D.208 J
Solution
ᐃQ = nCp ᐃT (Isobaric process)
= 2 ×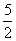 R × (35 - 30)
R × (35 - 30)
= 208J
= 2 ×
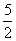 R × (35 - 30)
R × (35 - 30)= 208J
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The imstryment used to measure the temperature of the source from its thermal radiation is :...In an isothermal state, which of the following statements is true :-...A Carnot engine works between temperature 727oC and 27oC. The efficiency of the engine is :...Heat travels through vacuum by :...Water is used to cool the radiators of engines in cars because :...