ThermodynamicsHard
Question
Steam at 100o C is passes into 1.1 kg of water contained in a calorimeter of water equivalent 0.02 kg at 15oC till the temperature of the calorimeter and its content rises to 80o C . The mass of the steam condensed in king is
Options
A.0.130
B.0.065
C.0.260
D.0.135
Solution
Heat required Q = (1.1+ 0.02) × 103 × 1 × (80 -15)
= 72800 cal
Therefore, mass of steam condensed (in kg)
m =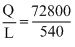 × 10-3 = 0.135 kg
× 10-3 = 0.135 kg
∴ Correct option is (d).
= 72800 cal
Therefore, mass of steam condensed (in kg)
m =
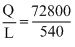 × 10-3 = 0.135 kg
× 10-3 = 0.135 kg ∴ Correct option is (d).
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The correct value of 0oC on kelvin scale is :...An ideal gas at 27o C is heated under constant pressure such that its volume is doubled, its temperature will be :...One mole of a monatomic ideal gas is taken along two cyclic processes E → F → G → E and E → F &#...An ideal gas at 27oC is compressed adiabatically to 8/27 of its original volume. If γ = 5/3, then the rise in tempe...The ratio of the speed of sound in nitrogen gas to that in helium gas, at 300 K is...