Chemical Kinetics and Nuclear ChemistryHard
Question
A radioactive element gets spilled over the floor of a room. Its half-life period is 30 days. If the initial activity is ten times the permissible value, after how many days will it be safe to enter the room?
Options
A.1000 days
B.300 days
C.10 days
D.100 days
Solution
Activity 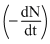 ∝ N
∝ N
N = No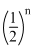
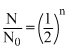
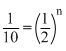 ⇒ 10 = 2n
⇒ 10 = 2n
log10 = nlog2
⇒ n =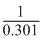 = 3.32
= 3.32
t = n × t112
= 3.32 × 30 = 99.6 days
Hence, (4) is correct.
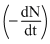 ∝ N
∝ NN = No
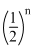
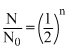
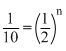 ⇒ 10 = 2n
⇒ 10 = 2nlog10 = nlog2
⇒ n =
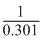 = 3.32
= 3.32 t = n × t112
= 3.32 × 30 = 99.6 days
Hence, (4) is correct.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
At 370 K a gaseous reaction X → 2Y + Z is found to be first order. Starting the reaction from pure X the total pre...The rate of a reaction increases four-fold when the concentration of reactant is increased 16 times. If the rate of reac...Match the graphical study with the order of the reactions : I : Rate II : Half life III : (a - x)-1...A reaction takes place in three steps having rate constants K1, K2, K3 respectively. The overall rate constant K = . If ...The energy equivalent of 2.0 mg mass defect is...