Chemical Kinetics and Nuclear ChemistryHard
Question
A reaction takes place in three steps having rate constants K1, K2, K3 respectively. The overall rate constant K = 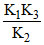 . If energies of activations for the three steps are 40, 30, 20 kJ respectively, the overal energy of activation is :
. If energies of activations for the three steps are 40, 30, 20 kJ respectively, the overal energy of activation is :
Options
A.10
B.15
C.30
D.60
Solution
given K = 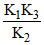
we know that for any reaction
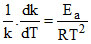
so differentiating the given relation of k with k1, k2 & k3 with respect to temperature, we have
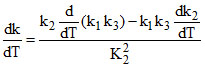 =
= 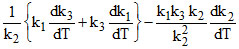
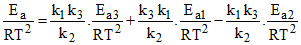
Ea = Ea3 + Ea1 - Ea2
= (20 + 40 - 30) = 30 kJ/mol
we know that for any reaction
so differentiating the given relation of k with k1, k2 & k3 with respect to temperature, we have
Ea = Ea3 + Ea1 - Ea2
= (20 + 40 - 30) = 30 kJ/mol
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
In the nuclear transmutation49Be + X → 48Be + Y (X, Y) is(are)...The rate of chemical reaction is directly proportional to :...2713Al is a stable element. 2913Al is expected to disintegrate by :...If I is the intensity of absorbed light and C is the concentration of AB for photochemical process. AB + hv → AB*,...In the following graphical representation for the reaction A → B there are two types of regions :...