Chemical Kinetics and Nuclear ChemistryHard
Question
At 370 K a gaseous reaction X → 2Y + Z is found to be first order. Starting the reaction from pure X the total pressure is observed 156 mm Hg after 15 minutes. After long time pressure is observed 240 mm Hg when X is completely dissociated. What will be the pressure of X after 15 minutes ?
Options
A.42 mm Hg
B.54 mm Hg
C.36 mm Hg
D.80 mm Hg
Solution
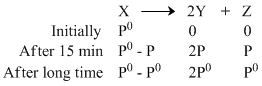
= 0
∵ 2P0 + P0 = 240 mm Hg
∴ P0 = 80 mm Hg
After 15 min P0 + 2P = 156 Hg
P = 38 mm Hg
Pressure of X after 15 min = P0 - P
= 42 mm Hg
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
As the initial concentration increases from 0.75 to 1.55 M in a reaction, t1/2 decreases from 60 to 29 s. The order of t...A reaction is catalysed by H+ ion;and in the rate law the dependence of rate is of first order with respect to the conce...Bi210 has a half - life of 5days. The time taken, for of a sample to decay, is :...In a first-order reaction, the activity of reactant drops from 800 mol/dm3 to 50 mol/dm3 in 2 × 104 s. The rate constant...The rate equation for the reaction 2A + B → C is found to be: rate k[A][B]. The correct statement in relation to t...