Chemical Kinetics and Nuclear ChemistryHard
Question
Match the graphical study with the order of the reactions :
I : Rate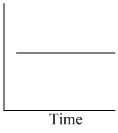 II : Half life
II : Half life  III : (a - x)-1
III : (a - x)-1 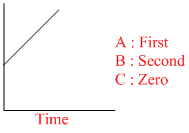
I : Rate
 II : Half life
II : Half life  III : (a - x)-1
III : (a - x)-1 
Options
A.I → A, II → B, III → C
B.I → B, II → C, III → A
C.I → C, II → B, III → A
D.I → C, II → A, III → B
Solution
Graph I - Rate independent of time, zero order Rxn.
Graph II - Half life independent of time, Ist order Rxn.
Graph III - between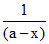 and time, straight line, second order Rxn.
and time, straight line, second order Rxn.
Graph II - Half life independent of time, Ist order Rxn.
Graph III - between
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
An aqueous solution is 1.00 molal in KI. Which change will cause the vapour pressure of the solution to increase ?...An endothermic reaction with high activation energy for the forward reaction is given by the diagram...Isogamous condition with non-flagallated gametesis found in :...During the kinetic study of the reaction, 2A + B → C + D, following results were obtained Based on the above data ...A reaction takes place in three steps having rate constants K1, K2, K3 respectively. The overall rate constant K = . If ...