Chemical Kinetics and Nuclear ChemistryHard
Question
The rate of a reaction increases four-fold when the concentration of reactant is increased 16 times. If the rate of reaction is 4 × 10-6 mol L-1 s-1 when the concentration of the reactant is 4 × 10-4 mol L-1, the rate constant of the reaction will be
Options
A.2 × 10-4 moil1/2 L-1/2 s-1
B.1 × 10-2 s-1
C. 2 × 10-4 mol-1/2 L1/2 s-1
D.25 mol-1 L min-1
Solution
Rate ∝ 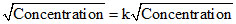
k =
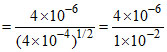 = 2 × 10-4 mol1/2 L-1/2 s-1
= 2 × 10-4 mol1/2 L-1/2 s-1
k =
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Which of the following ions will have the minimum coagulating value for the sol obtained by adding FeCl3 solution to sli...In the following graphical representation for the reaction A → B there are two types of regions :...Correct order of polarising power is :-...If I is the intensity of absorbed light and C is the concentration of AB for photochemical process. AB + hv → AB*,...The rate of a reaction doubles when its temperature changes from 300 K to 310 K. Activation energy of such a reaction wi...