Gaseous StateHard
Question
Equal masses of methane and oxygen are mixed in an empty container at 25oC. The fraction of the total pressure exerted by oxygen is
Options
A.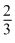
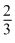
B.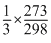
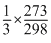
C.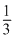
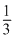
D.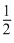
Solution
Let the mass of methane and oxygen is w
mole fraction of oxygen =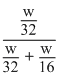
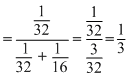
Let the total pressure be P
The pressure exerted by oxygen (partial pressure) = XO2 × Ptotal
⇒ P ×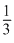
Hence, (3) is correct.
mole fraction of oxygen =
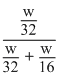
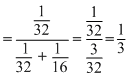
Let the total pressure be P
The pressure exerted by oxygen (partial pressure) = XO2 × Ptotal
⇒ P ×
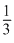
Hence, (3) is correct.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
1 mol of a gaseous aliphahatic compound CnH3nOm is completely burnt in an excess of oxygen. The contraction in volume is...Equal weight of SO2 and O2 are mixed in an container at 25oC. The fraction of the total pressure exerted by O2 is :-...Which of the following expression correctly represents the relationship between the average kinetic energy of CO and N2 ...Equal weights of methane and hydrogen are mixed in an empty container at 25oc. The fraction of the total pressure exerte...The inversion temperature of hydrogen is (given van der Waal′s constants a and b are 0.244 atmL2mol-2 and 1.027 L ...