Gaseous StateHard
Question
Which of the following expression correctly represents the relationship between the average kinetic energy of CO and N2 molecules at the same temperature.
Options
A.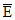 (CO) >
(CO) > 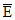 (N2)
(N2)
B.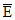 (CO) <
(CO) < 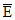 (N2)
(N2)
C.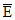 (CO) =
(CO) = 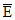 (N2)
(N2)
D.Cannot be predicted unless volumes of the gases are given
More Gaseous State Questions
If Z is the number of atoms in the unit cell that represents the closest packing sequence..... ABC ABC ......, the numbe...1 mol of an ideal gas at 27oC is compressed reverssibly from 20 L to 2L. Find out ᐃS :-...On the surface of the earth at 1 atm pressure, a balloon filled with H2 gas occupies 500 mL. This volume is 5/6 of its m...For one mole of a van der Waals gas when b = 0 and T = 300 K, the PV vs. 1/V plot is shown below. The value of the van d...If the pressure of a gas contained in a closed vessel is increased by 0.4 % when heated by 1oC its initial temperature m...