Gaseous StateHard
Question
The inversion temperature of hydrogen is (given van der Waal′s constants a and b are 0.244 atmL2mol-2 and 1.027 L mol-1 respectively)
Options
A.440
B.220
C.110
D.330
Solution
Gases become cooler during joule Thomson′s expansion only if they are below acertain temperature known as inversion temperature (Ti). The inversion temperature is characteristic of each gas is given by
Ti =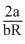
Where R is gas constant
Given a = 0.244 atmL2mol-2
b = 0.027 L mol-1
R = 0.0821 L atmdeg-1 mol-1
∴ Ti =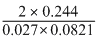 = 220K
= 220K
Ti =
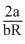
Where R is gas constant
Given a = 0.244 atmL2mol-2
b = 0.027 L mol-1
R = 0.0821 L atmdeg-1 mol-1
∴ Ti =
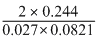 = 220K
= 220KCreate a free account to view solution
View Solution FreeMore Gaseous State Questions
If most probable velocity is represented by ′a′ and fraction possessing it by ′f′, then with inc...The volume of CO2 produced by the combustion of 40 ml of gaseous acetone in excess of oxygen is...Helium atom is two times heavier than a hydrogen molecule. At 298 K, the average kinetic energy of a helium atom is...When does a gas deviate the most from its ideal behaviour ?...Equal weights of ethane & hydrogen are mixed in an empty container at 25oC, the fraction of the total pressure exerted b...