Gaseous StateHard
Question
Equal weights of methane and hydrogen are mixed in an empty container at 25oc. The fraction of the total pressure exerted by hydrogen is :
Options
A.

B.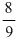
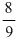
C.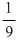
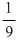
D.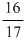
Solution
Fraction of total pressure exerted by H2 = mole fraction of H2
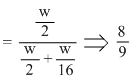
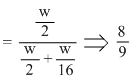
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
The average velocity of an ideal gas molecule at 27oC is 0.3/s. The avarage velocity at 927oC will be :...A chemist has synthesized a greenish yellow gaseous compound of chlorine and oxygen and finds that its density is 7.71 g...For gaseous state, if most probable speed is denoted by C*, average speed by and mean square speed by C, then for a larg...For a real gas the P-V curve was experimentally plotted and it had the following appearance. With respect to liquifactio...2.5 L of a sample of a gas at 27oC and 1 bar pressure is compressed to a volume of 500 mL keeping the temperature consta...