Gaseous StateHard
Question
1 mol of a gaseous aliphahatic compound CnH3nOm is completely burnt in an excess of oxygen. The contraction in volume is (assume water get condensed out)
Options
A.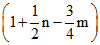
B.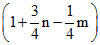
C.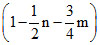
D.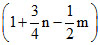
Solution
CnH3nOm + yO2 → nCo2 (g) + 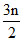 H2O(l)
H2O(l)
Contraction in volume = Contraction in moles of gas = 1 +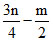
Contraction in volume = Contraction in moles of gas = 1 +
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
A real gas most closely approaches the behaviour of an ideal gas at -...By what factor does the average velocity of a gaseous molecule increase when the temperature (in Kelvin) is doubled ?...At Boyle′s temperature, the value of compressibility factor Z = (PVm / RT = Vreal/Videal) has a value of 1, over a...Select correct statement(s):...If most probable velocity is represented by ′a′ and fraction possessing it by ′f′, then with inc...