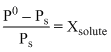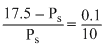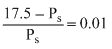SolutionHard
Question
The vapour pressure of water at 20oC is 17.5 mm Hg. If 18 g of glucose (C6H12O6) is added to 178.2 g of water at 20oC, the vapour pressure of the resulting solution will be
Options
A.17.675 mm Hg
B.15.750 mm Hg
C.16.500 mm Hg
D.17.325 mm Hg
More Solution Questions
Which relations are not correct for an aqueous dilute solution of K3PO4 if its degree of dissociation is α ?...A complex containing K+, Pt(IV) and Cl- is 100% ionised giving i = 3. Thus, complex is :...The phase diagrams for the pure solvent (solid lines) and the solution (non-volatile solute, dashed line) are recorded b...If relative decrease in vapour pressure is 0.4 for a solution containing 1 mol NaCl in 3 mol H2O, NaCl is .... % ionised...Which statement about the triple point of a substance is/are incorrect...