SolutionHard
Question
If relative decrease in vapour pressure is 0.4 for a solution containing 1 mol NaCl in 3 mol H2O, NaCl is .... % ionised.
Options
A.60%
B.50%
C.100%
D.40%
Solution
RLVP = 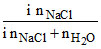
0.4 =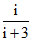
so i = 2
∵ i = 1 + α so α = 1 or 100%
0.4 =
so i = 2
∵ i = 1 + α so α = 1 or 100%
Create a free account to view solution
View Solution FreeMore Solution Questions
A living cell contains a solution which is isotonic with 0.2 M glucose solution. What osmotic pressure develops when the...The freezing point depression constant for water is 1.86oC. If 5.00 g Na2SO4 is dissolved in 45.0 g H2O, the freezing po...Low concentration of oxygen in blood and tissues of people living at high altitude is due to...20 g of a binary electrolyte (mol. mass = 100) are dissolved in 500 g of water. The freezing point of the solution is -0...Which of the following combinations are correct for a binary solution, in which the solute as well as solvent are liquid...