SolutionHard
Question
Which relations are not correct for an aqueous dilute solution of K3PO4 if its degree of dissociation is α ?
Options
A.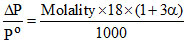
B.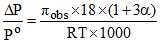
C.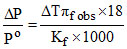
D.Mw of K3PO4 = Mwobs × (1 + 3α)
Solution
For electrolyte
(Mw1 = 18 for H2O)
Also, pobs = C × R × T (1 + 3α)
∴
ᐃTf obs = Kf × molality × (1 + 3α)
i = (1 + 3α) =
Therefore, molecular weight of K3PO4 = Mobs × (1 + 3α)
Create a free account to view solution
View Solution FreeMore Solution Questions
Thevan′t Hoff factor, i for a compound which undergoes dissociation in one solvent and association in other solven...For an ideal solution of A and B, YA is the mole fraction of A in the vapour phase at equilibrium. Which of the followin...An amount of 1 mole of a non-volatile solid is dissolved in 200 moles of water. The solution is cooled to a temperature ...The depression of freezing points of 0.05 molal aqueous solution of the following compounds are measured.1. NaCl 2. K2SO...What will be the osmotic pressure of decimolar solution of glucose at 30°C?...