SolutionHard
Question
The phase diagrams for the pure solvent (solid lines) and the solution (non-volatile solute, dashed line) are recorded below :
The quantity indicated by L in the figure is :
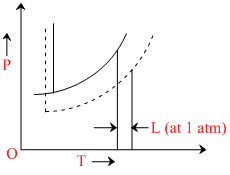
The quantity indicated by L in the figure is :

Options
A.ᐃp
B.ᐃTf
C.Kbm
D.Kfm
Solution
L indicates elevation in boiling pt. i.e., kbm.
Create a free account to view solution
View Solution FreeMore Solution Questions
Some entropy change are represented in figure. Select correct entropy change....The ebullioscopic constant of a liquid solvent is the elevation of boiling point of...Which of the following curves represents the Henry′s law ?...Depression of freezing point of 0.01 molal aq. CH3COOH solution is 0.02046o. 1 molal urea solution freezes at - 1.86°C. ...Assuming each salt to be 90% dissociated which of the following will have highest osmotic pressure ?...