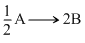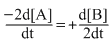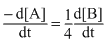Chemical Kinetics and Nuclear ChemistryHard
Question
For a reaction 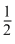 A → 2B, rate of disappearance of ′A′ is related to the rate of appearance of ′B′ by the expression
A → 2B, rate of disappearance of ′A′ is related to the rate of appearance of ′B′ by the expression
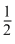 A → 2B, rate of disappearance of ′A′ is related to the rate of appearance of ′B′ by the expression
A → 2B, rate of disappearance of ′A′ is related to the rate of appearance of ′B′ by the expressionOptions
A.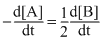
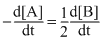
B.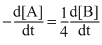
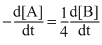
C.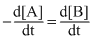
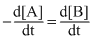
D.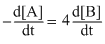
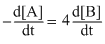
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The rate of a reaction increases four-fold when the concentration of reactant is increased 16 times. If the rate of reac...Inversion of sucrose (C12H22O11) is first-order reaction and is studied by measuring angle of rotation at different inst...How many coulomb of electricity would be required to reduce the iron in 32.9 g of potassium hexa cyano ferrate (III) K3F...A reaction involving two different reactants can never be...The half life period of a first order chemical reaction is 6.93 minutes. The time required for the completion of 99% of ...