Chemical Kinetics and Nuclear ChemistryHard
Question
Inversion of sucrose (C12H22O11) is first-order reaction and is studied by measuring angle of rotation at different instant of time
C12H22O11 + H2O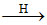 C6H12O6
C6H12O6
Sucrose Clucose Fructose
d d l
If (r∞ - r0) = a and (r∞ - rt) = (a - x) (where r0, rt and r∞ are the angle of rotation at the start, at the time t and at the end of the reaction respectively, then there is 50% inversion when :
C12H22O11 + H2O
Sucrose Clucose Fructose
d d l
If (r∞ - r0) = a and (r∞ - rt) = (a - x) (where r0, rt and r∞ are the angle of rotation at the start, at the time t and at the end of the reaction respectively, then there is 50% inversion when :
Options
A.r0 = 2rt - r∞
B.r0 = rt - r∞
C.r0 = rt - 2r∞
D.r0 = rt + r∞
Solution
Given (r∞ - r0) = a , (r∞ - rt) = (a - x)
At 50% Inversion
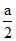 = (a - x)
= (a - x)
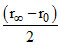 = (r∞ - r0)
= (r∞ - r0)
(r∞ - r0 ) = 2r∞ - 2rt
r0 = 2rt - r∞
At 50% Inversion
(r∞ - r0 ) = 2r∞ - 2rt
r0 = 2rt - r∞
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For a reaction A 2B starting with 1 M of ′A′ only, concentration of B (in M) after 100 sec. and 200 sec. is ...Consider the chemical reaction,N2(g) + 2H2(g) → 2NH3(g) The rate of this reaction can be expressed in terms of tim...For the second-order reaction A + B → Products, the rate constant k, is given as$k = \frac{2.303}{(a - b)t}\log\frac{b(a...Bombardment of aluminium by a-particle leads to its artificial disintegration in two ways, (i) and (ii) as shown. Produc...Determine the average life of U238 having t0.5 = 138.5 day....