Chemical Kinetics and Nuclear ChemistryHard
Question
How many coulomb of electricity would be required to reduce the iron in 32.9 g of potassium hexa cyano ferrate (III) K3Fe(CN)6 to metallic iron ?
(at mass of K = 39, Fe = 56)
(at mass of K = 39, Fe = 56)
Options
A.289500 C
B.28950 C
C.9650 C
D.19300 C
Solution
Iron in the complex is Fe3+
K3Fe(CN)6 + 3e- → 3K+ 6CN- + Fe
1 mol 3 F
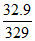 = 0.1 mol 0.3 F
= 0.1 mol 0.3 F
K3Fe(CN)6 + 3e- → 3K+ 6CN- + Fe
1 mol 3 F
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Sulphide ores of metals are usually concentrated by froth floatation process. Which one of the following sulphide ores i...Which of the following radioisotopes is used as anticancerous ?...Number of Faraday required to liberate 8 g. of H2 is :-...For the non-stoichiometre reaction 2A + B → C + D, the following kinetic data were obtained in three separate expe...The Golgi complex plays a major role :...