Chemical Kinetics and Nuclear ChemistryHard
Question
The half life period of a first order chemical reaction is 6.93 minutes. The time required for the completion of 99% of the chemical reaction will be (log 2 = 0.301) :
Options
A.230.3 minutes
B.23.03 minutes
C.46.06 minutes
D.460.6 minutes
Solution
∵ λ = 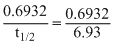 min-1
min-1
Also t =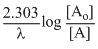
[Ao] = initial concentration (amount)
[A] = final concentration (amount)
∴ t =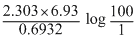
= 46.06 minutes
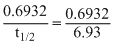 min-1
min-1 Also t =
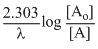
[Ao] = initial concentration (amount)
[A] = final concentration (amount)
∴ t =
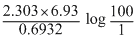
= 46.06 minutes
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Decay constant of a radioactive substance is 69.3sec-1, find t1/16 of the same substance....Consider an endothermic reaction, X → Y with the activation energies Eb and Ef for the backward and forward reacti...Identify the correct statement when CuSO4(aq) is mixed with KI(aq) :-...If no catalyst (H+) is present in acid hydrolysis of ester (in above question) then rate constant K is :...For the reaction2N2O5 → 4NO2 + O2The rate of reaction is :...