JEE Main | 2014ElectrochemistryHard
Question
The metal that cannot be obtained by electrolysis of an aqueous solution of its salts is
Options
A.Ag
B.Ca
C.Cu
D.Cr
Solution
On electrolysis only in case of Ca2+ salt aqueous solution H2 gas discharge at Cathode.
Case of Cr
At cathode : Cr3+ + 2e- → Cr
So, Cr is deposited.
Case of Ag
At cathode : Ag+ + e- → Ag
So, Ag is deposited.
Case of Cu
At cathode : Cu2+ + 2e- → Cu
Case of Ca2+
At cathode : H2O + e- →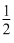 H2 + OH-
H2 + OH-
Case of Cr
At cathode : Cr3+ + 2e- → Cr
So, Cr is deposited.
Case of Ag
At cathode : Ag+ + e- → Ag
So, Ag is deposited.
Case of Cu
At cathode : Cu2+ + 2e- → Cu
Case of Ca2+
At cathode : H2O + e- →
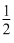 H2 + OH-
H2 + OH-Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
What is the emf at 25oC for the cell, Ag Pt The standard reduction potentials for the half-reactions AgBr + e- → A...Indicator electrode is...The molar conductivities ∧oNaOAc and ∧oHCl at infinite dilution in water at 25oC are 91.0 and 426.2 S cm2/mo...The reaction, H2(g) + AgCl(s) → H+ (aq) + Cl- (aq) + Ag(s) occurs in the galvanic cell :...Emf for the cell reaction Zn|Zn2+(0.001M)||Zn2+(0.1M)|Zn...