ElectrochemistryHard
Question
Emf for the cell reaction Zn|Zn2+(0.001M)||Zn2+(0.1M)|Zn
Options
A.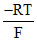 ln(0.01)
ln(0.01)
B.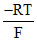 ln(0.1)
ln(0.1)
C.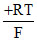 ln(0.01)
ln(0.01)
D.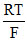 ln(0.1)
ln(0.1)
Solution
Ecell = Eocell - 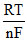 lnQ
lnQ
= Eocell -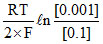
= Eocell -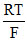 ln (10-2)1/2
ln (10-2)1/2
= 0 -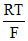 ln (10-1)
ln (10-1)
Ecell = -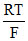 ln[0.1]
ln[0.1]
= Eocell -
= Eocell -
= 0 -
Ecell = -
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
In hydrogen - oxygen fuel cell, combustion of hydrogen occurs to...The electric charge for electrode deposition of one gram equivalent of a substance is :...Molar conductivities at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively. ...Which statement is correct....Both oxidation and reduction takes place in :...