ElectrochemistryHard
Question
The reaction,  H2(g) + AgCl(s) → H+ (aq) + Cl- (aq) + Ag(s) occurs in the galvanic cell :
H2(g) + AgCl(s) → H+ (aq) + Cl- (aq) + Ag(s) occurs in the galvanic cell :
 H2(g) + AgCl(s) → H+ (aq) + Cl- (aq) + Ag(s) occurs in the galvanic cell :
H2(g) + AgCl(s) → H+ (aq) + Cl- (aq) + Ag(s) occurs in the galvanic cell :Options
A.Ag | AgCl(s)| KCl(soln) || AgNO3 | Ag
B.Pt | H2 (g) | HCl(soln) || AgNO3(soln) | Ag
C.Pt | H2 (g) | HCl(soln) || AgCl(s) | Ag
D.Pt | H2 (g) | KCl(soln) || AgCl(s) | Ag
Solution
In this cell H2 is oxidised to H+ and acts as anode while Ag+ is reduced to Ag. The cell may be represented as
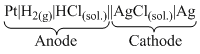
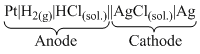
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A current of 3.7 A is passed for 6 h between nickel electrodes in 0.50 L of 2 M solution of Ni(NO3)2. The molarity of Ni...A current of 3 A was passed for 1 hour through an electrolyte solution of AxBy in water. If 2.977 g of A (atomic weight ...An electrolytic cell contains a solution of Ag2SO4 and platinum electrodes. A current is passed until 1.6 g of O2 has be...When the electric current is passed through a cell having an electrolyte, the positive ions move towards cathode and neg...When an electric current is drawn from a galvanic cell...