ElectrochemistryHard
Question
What is the emf at 25oC for the cell, Ag 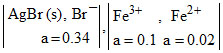 Pt
Pt
The standard reduction potentials for the half-reactions AgBr + e- → Ag + Br- and Fe3+ + e- → Fe2+ are + 0.0713 V and + 0.770 V respectively.
The standard reduction potentials for the half-reactions AgBr + e- → Ag + Br- and Fe3+ + e- → Fe2+ are + 0.0713 V and + 0.770 V respectively.
Options
A.0.474 volt
B.0.529 volt
C.0.356 volt
D.0.713 volt
Solution
Ecell = (0.77 - 0.0713) - 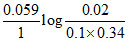 = 0.713 volt.
= 0.713 volt.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Which of the following is/are function(s) of salt - bridge ?...Calculate the quantity of electricity that would be required to reduce 12.3 g of nitrobenzene to aniline if the current ...A volume of 100 ml of a buffer of 1 M – NH3 and 1 M – NH4+ is placed in two half-cells connected by a salt bridge. A cur...The potential of the Daniell cell, Zn Cu was reported by Buckbee, Surdzial and Metz as Eo = 1.1028 - 0.641 × 10-3 T...An aqueous solution of X is added slowly to an aqueous solution of Y as shown in List-I. The variation in conductivity o...