ThermochemistryHard
Question
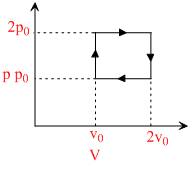
The above p-v diagram represents the thermodynamic cycle of an engine, operating with an ideal monoatomic gas. The amount of heat, extracted from the source in a single cycle is :
Options
A.p0v0
B.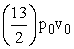
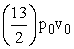
C.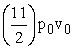
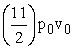
D.4p0v0
Solution
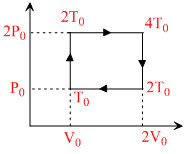
heat supplied = nCv(2T0 - T0) + nCp (4T0 - 2T0)
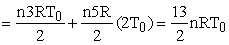
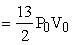
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
The potential energy diagram for a reaction R   → P is give below ᐃHo of the reaction corresponds to the...A coffee cup calorimeter initially contains 125 g of water, at a temperature of 24.20C. 8g of ammonium nitrate (NH4NO3),...When 1.0 g of oxalic acid is burnt in a bomb calorimeter whose heat capacity is 8.75 kJ/K, the temperature increases by ...In which of the following the hydration energy is higher than the lattice energy :-...Combustion of glucose takes place according to the equation :C6H12O6 + 6O2 → 6CO2 + 6H2O, ᐃH = - 72 kcalThe ...