ThermochemistryHard
Question
Combustion of glucose takes place according to the equation :
C6H12O6 + 6O2 → 6CO2 + 6H2O, ᐃH = - 72 kcal
The energyrequired for production of 1.6g of glucose is [molecular mass of glucose is 180 gm]
C6H12O6 + 6O2 → 6CO2 + 6H2O, ᐃH = - 72 kcal
The energyrequired for production of 1.6g of glucose is [molecular mass of glucose is 180 gm]
Options
A.0.064 kcal
B.0.64 kcal
C.6.4 kcal
D.64 kcal
Solution
Formation of glucose will be the given by reaction taking place reverse direction.
6CO2 + 6H2P + 72Cal → C6H12O6 + 6O2 For production of 180 gm of glucose, heat energy is 72 k cals.
So required heat for 1.6 gm of glocose =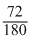 × 1.6 = 0.64 kcal
× 1.6 = 0.64 kcal
6CO2 + 6H2P + 72Cal → C6H12O6 + 6O2 For production of 180 gm of glucose, heat energy is 72 k cals.
So required heat for 1.6 gm of glocose =
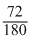 × 1.6 = 0.64 kcal
× 1.6 = 0.64 kcalCreate a free account to view solution
View Solution FreeMore Thermochemistry Questions
For the allotropic change represented by the equation C(graphite) → C(diamond); ΔH = +1.9 kJ. If 6.0 g of diamond and 6....Use the following data to calculate the enthalpy of hydration for caesium iodide and caesium hydroxide, respectively.Com...The standard enthalpies of formation of H2O(l), Li+ (aq) and OH− (aq) are −285.8, −278.5 and −228.9 kJ/mol, respectively...Determine ᐃUo at 300 K for the following reaction using the listed enthalpies of reaction :4CO(g) + 8H2(g) →...The thermochemical equation for the dissociation of hydrogen gas into atoms may be written asH2 → 2H; ΔH = 432.0 kJ.What...