ThermochemistryHard
Question
The potential energy diagram for a reaction R → P is give below
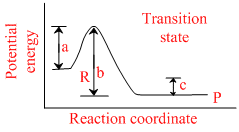
ᐃHo of the reaction corresponds to the energy :

ᐃHo of the reaction corresponds to the energy :
Options
A.a
B.b
C.c
D.a + b
Solution
Activation energy of forward reaction = a
Activation energy of backward reaction = a
∴ ᐃH = b - a
Since b > a, there fore reaction is exothermic
Activation energy of backward reaction = a
∴ ᐃH = b - a
Since b > a, there fore reaction is exothermic
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
The enthalpy change for a reaction does not depend upon...The enthalpy changes for the following processes are listed below: Cl2(g) = 2Cl(g), 242.3 kJ mol-1 I2(g) = 2I(g), 151.0 ...The factor of ΔG values is important in metallurgy. The ΔG values for the following reactions at 800o C are given as fol...The thermochemical equation for the dissociation of hydrogen gas into atoms may be written asH2 → 2H; ΔH = 432.0 kJ.What...The difference between enthalpies of reaction at constant pressure and constant volume for the reaction2C6H6(l) + 15O2(g...