ThermochemistryHard
Question
A coffee cup calorimeter initially contains 125 g of water, at a temperature of 24.20C. 8g of ammonium nitrate (NH4NO3), also at 24.2oC, is added to the water, and the final temperature is 18.2oC. what is the heat of solution of ammonium nitrate in kJ/mol ? The specific heat capacity of the solution is 4.2 J/oC g.
Options
A.33.51 kJ/mol
B.39.5 kJ/mol
C.32.2 kJ/mol
D.37.3 kJ/mol
Solution
áƒHsolution = 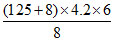 × 80 = 33516 J/mol or 33.51 kJ/mol.
× 80 = 33516 J/mol or 33.51 kJ/mol.
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
The enthalpy of neutralization of a strong acid by a strong base is −57.32 kJ mol–1. The enthalpy of formation of water ...The Δf Ho for CO2(g), CO(g) and H2O(g) are −393.5, −110.5 and −241.8 kJ mol–1, respectively. The standard enthalpy chang...Study the following thermochemical equations.A → B; ΔH = +100 kcal B → C; ΔH = −80 kcalThe correct order of enthalpies o...Calculate the heat effect produced when a solution of 1 mole of ethanol in 5 moles of water is mixed with a solution of ...The standard heat of formation of carbon atom in gaseous state in kJ/mol is - áƒHo (atomisation of CO gas) = 1072 kJ/mo...