ThermochemistryHard
Question
When 1.0 g of oxalic acid is burnt in a bomb calorimeter whose heat capacity is 8.75 kJ/K, the temperature increases by 0.312 K. The enthalpy of combustion of oxalic acid at 27oC is :-
Options
A.-245.7 kJ/mol
B.-244.452 kJ/mol
C.-246.947 kJ/mol
D.None of these
Solution
H2C2O4(l) + 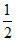 O2(g) → H2O(l) + 2CO2(g)
O2(g) → H2O(l) + 2CO2(g)
ᐃng =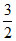
ᐃu =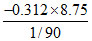 = -245.7 kJ/mol
= -245.7 kJ/mol
ᐃH = ᐃu + ᐃngRT
= - 245.7 +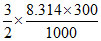
= - 246.947 kJ/mol
ᐃng =
ᐃu =
ᐃH = ᐃu + ᐃngRT
= - 245.7 +
= - 246.947 kJ/mol
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
Given the following equations and ᐃHo values, determine the enthalpy of reaction at 298 K for the reaction : C2H4 ...Which of the following is arranged in the increasing order of enthalpy of vaporisation ?...The enthalpies of formation of FeO(s) and Fe2O3(s) are −65.0 and −197.0 kcal/mol, respectively. A mixture of the two oxi...The standard enthalpy of formation of NH3 is - 46.0 kJ mol-1. If the enthalpy of formation of H2 from its atoms is - 436...The dissolution of CaCl2·6H2O in a large volume of water is endothermic to the extent of 3.5 kcal/mol. For the reaction,...