Chemical Kinetics and Nuclear ChemistryHard
Question
The rate constant k, for the reaction N2O5(g) → 2NO2(g) + 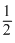 O2(g) is 1.3 × 10-2s-1. Which equation given below describes the change of [N2O5]0 and [N2O5]t correspond to concentration of N2O5 initially and at time t.
O2(g) is 1.3 × 10-2s-1. Which equation given below describes the change of [N2O5]0 and [N2O5]t correspond to concentration of N2O5 initially and at time t.
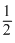 O2(g) is 1.3 × 10-2s-1. Which equation given below describes the change of [N2O5]0 and [N2O5]t correspond to concentration of N2O5 initially and at time t.
O2(g) is 1.3 × 10-2s-1. Which equation given below describes the change of [N2O5]0 and [N2O5]t correspond to concentration of N2O5 initially and at time t.Options
A.[N2O5]t = [N2O5]0 + kt
B.[N2O5]0 = [N2O5]t ekt
C.log[N2O5]0 = log [N2O5]t + kt
D.In 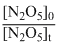 = kt 1
= kt 1
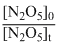 = kt 1
= kt 1Solution
As the unit of rate constant is sec-1, so the reaction is first roder reaction. Hence k = 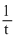 = log
= log 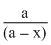 or kt = log
or kt = log 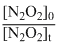
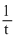 = log
= log 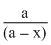 or kt = log
or kt = log 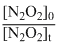
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The half-life period of a radioactive element is 140 days. After 560 days, one gram of the element will reduce to :...The physical adsorption of gases on the solid surface is due to:...Which of the following statement is wrong about the rate of reaction?...For a given reaction, the concentration of the reactant plotted against time gave a straight line with negative slope. T...On increasing the temperature, the half-life of reactions...