Chemical Kinetics and Nuclear ChemistryHard
Question
A(g) → B(g) + C(g)
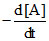 = k [A]
= k [A]
At the start pressure is 100 mm and after 10 min , pressure is 120 mm. hence , rate constant (min-1) is :
At the start pressure is 100 mm and after 10 min , pressure is 120 mm. hence , rate constant (min-1) is :
Options
A.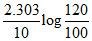
B.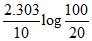
C.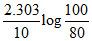
D.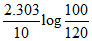
Solution
A(g) → B(g) + C(g)
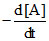 = k [A]
= k [A]
A (g) → B (g) + C (g)
100 0 0 t = 0
(100 - P) P P t = 10 min. (feuV)
100 - P + P + P = 120
(100 + P) = 120
Then
k =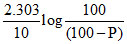
k =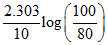
A (g) → B (g) + C (g)
100 0 0 t = 0
(100 - P) P P t = 10 min. (feuV)
100 - P + P + P = 120
(100 + P) = 120
Then
k =
k =
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
raph between log k and 1/T (k is rate constant in s-1 and T is the temperature in k) is a straight line if OX = 5 and sl...The correct difference between first- and second-order reaction is that...Which of the following graphs represents zero order if A → PAt t = 0 ⇒ [A]0At t = 0 ⇒ [A]t...At a certain temperature, the first order rate constant k1 is found to be smaller than the second order rate constant k2...Which of the following molecules is most suitable to disperse benzene in water ?...