Chemical BondingHard
Question
The heat of atomization of methane and ethane are 360 kJ/mol and 620 kJ/mol, respectively. The longest wavelength of light capable of breaking the C-C bond is (A vogadro number = 6.02 × 1023, h = 6.62 × 10-34 J s):
Options
A.2.48 × 103 nm
B.1.49 × 103 nm
C.2.49 × 104 nm
D.2.48 × 104 nm
Solution
CH4(g) → C(g) + 4H(g)
⇒ 4 × EC - H = 360 KJ/Mol. ⇒ EC - H = 90 KJ/Mol.
and C2H6(g) → 2C(g) + 6H(g)
⇒ EC - C + 6 × 90 = 620 ⇒ EC - C = 80 kJ/mol
⇒ NA ×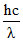 = 80 × 1000 J
= 80 × 1000 J
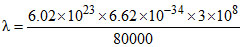
= 14.9 × 10-7 m = 1.49 × 10-6 m
= 1.49 × 103 nm.
⇒ 4 × EC - H = 360 KJ/Mol. ⇒ EC - H = 90 KJ/Mol.
and C2H6(g) → 2C(g) + 6H(g)
⇒ EC - C + 6 × 90 = 620 ⇒ EC - C = 80 kJ/mol
⇒ NA ×
= 14.9 × 10-7 m = 1.49 × 10-6 m
= 1.49 × 103 nm.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
On hybridisation of one s and one p-orbital we get :...Consider the plots, gives below, for the types of reaction:- nA → B + C (I) (II) (III)These plots respectively cor...The formal changes on the atoms marked as (1) to (4) in the Lewis representation of ${HnO}_{3}$ molecule respectively ar...Predict the correct intermediate and product in the following reaction :H3C-C≡CH→HgSO4H2O, H2SO2Interme...Given below are two statements:Statement I: The number of pairs among [ ${SiO}_{2},{CO}_{2}$ ], $\left\lbrack SnO,{SnO}_...