Chemical BondingHard
Question
Consider the plots, gives below, for the types of reaction:-
nA → B + C
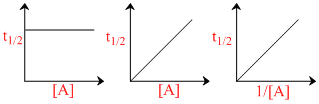
(I) (II) (III)
These plots respectively correspond to the reaction orders:
nA → B + C

(I) (II) (III)
These plots respectively correspond to the reaction orders:
Options
A.0, 1, 2
B.1, 2, 0
C.1, 0, 2
D.None of these
More Chemical Bonding Questions
On hybridisation of one s and one p-orbital we get :...If ᐃEN is 0.5 then percentage covalent character will be : (app.)...Which of the following is/are correct :-(a) Mg(HCO3)2 is responsible for permanent hardness(b) H2O2 is behave as oxidisi...The structure of IF7 is :...Among KO2, AlO2-, BaO2 and NO2+, unpaired electron is present in :...