Chemical BondingHard
Question
On hybridisation of one s and one p-orbital we get :
Options
A.two mutually perpendicular orbitals
B.two orbitals at 180o
C.for orbitals directed tetrahedrally
D.therr ofbitals in a plane
Solution
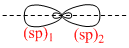
It will give two sp-hybrid orbitals which eill be arranged at 180o

Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Number of sigma bonds in P4O10 is...Which of the following is least likely to behave as Lewis base ?...What is the hybridisation of anionic part of product when XeF2 and PF5 are combined :...The tendency of BF3, BCl3 and BBr3 to behave as Lewis acid decreases in the sequence...The variation of extent of adsorption with pressure at a given constant temperature is given in following figure :Which ...